|
8/13/2023 0 Comments C2h4 molecular geometry bond angle 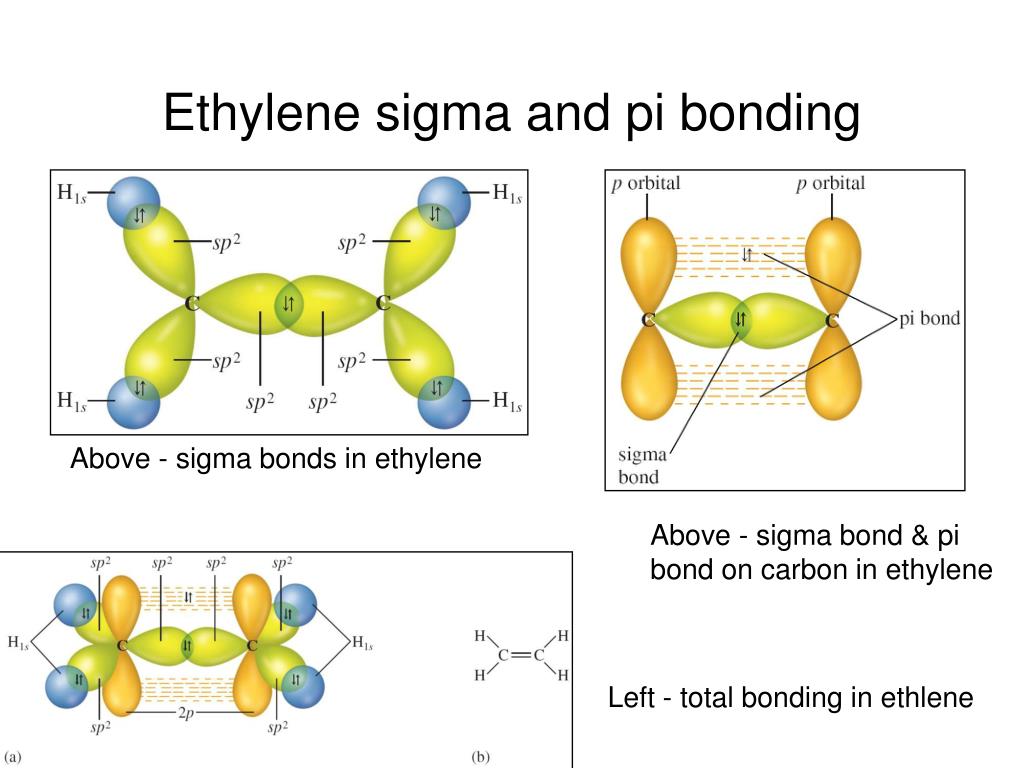
In the case of ethene, there is a difference from, say, methane or ethane, because each carbon is only joining to three other atoms rather than four. It is important that you have first met the idea of hybridisation in the more simple methane case. Use the BACK button on your browser to come back here when you have finished. Note: If you haven't read about bonding in methane, follow this link before you go any further. The carbon atom is now said to be in an excited state. The extra energy released when these electrons are used for bonding more than compensates for the initial input. There is only a small energy gap between the 2s and 2p orbitals, and an electron is promoted from the 2s to the empty 2p to give 4 unpaired electrons. 
This is exactly the same as happens whenever carbon forms bonds - whatever else it ends up joined to. The carbon atom doesn't have enough unpaired electrons to form the required number of bonds, so it needs to promote one of the 2s 2 pair into the empty 2p z orbital. Each line in this diagram represents one pair of shared electrons.Įthene is actually much more interesting than this.Įthene is built from hydrogen atoms (1s 1) and carbon atoms (1s 22s 22p x 12p y 1). You may also find it useful to read the article on orbitals if you aren't sure about simple orbital theory.Īt a simple level, you will have drawn ethene showing two bonds between the carbon atoms. It does not store any personal data.Important! You will find this much easier to understand if you first read the article about the bonding in methane. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. H2O- Bond angles are less than 109.5 degrees (103.9 as analyzed with chime on the class website) which is expected from this molecule’s hybridization. The carbon is sp hybridized and oxygens are sp2 hybridized. There is a double bond between each oxygen and carbon. What is the approximate bond angle in CO2?ĬO2- Bond angles are 180 degrees. As a result of the double bond C 2 H 2 molecular geometry is linear with a bond angle of 180 o.Ī triple bond is the bond that contain one sigma bond and two Pi bond, Acetylene (C2H2) is unsaturated with 2 carbon atoms that are bonded together in a triple bond. Given the distances between 3 atoms, one simple method for calculating bond angles is by use of the trigonometric cosine rule: cosγ = (A2 + B2 − C2) / 2 AB where A, B, C are the lengths of the sides of the triangle ABC, and γ is the angle A-C-B.Įthyne has a triple bond between the two carbon atoms. (A) The structural model for C2H4 consists of a double covalent bond between the two carbon atoms and single bonds to the hydrogen atoms. The 2s orbital in each carbon hybridizes with one of the 2p orbitals and forms two sp hybrid orbitals. In the formation of C2H2, the carbon atom needs extra electrons to form 4 bonds with hydrogen and other carbon atoms. These two orbitals form two pi bonds that result in the formation of triple bonds between carbon atoms. There are two-half filled 2p orbitals for each Carbon atom. Why is C2H2 a triple bond?Ĭ2H2 Hybridization The 1s orbital of the Hydrogen atom overlaps with the Carbon atom’s 2p orbital atom, making it an sp hybridization. Each H-C-H angle is 109.5° and each H-C-C angle is 109.5°. The two carbons are bonded together, and each is bonded to three hydrogens. What is the bond angle of C2H6?Įthane, C2H6, has a geometry related to that of methane. Moreover, carbon is bonding to carbon which gives acetylene (C2H2) a linear structure and a bond angle of 180°. Molecular Geometry of Acetylene (C2H2) Acetylene (C2H2) is a tetra atomic molecule having two different atoms bonding in equal numbers. What is the bond angle of c2h4?ĬCl4 has a tetrahedral geometry with bond angles of 109.5 °. 6 Which type of bond is present in C2H2?.1 How do you find the bond angle of C2H2?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
